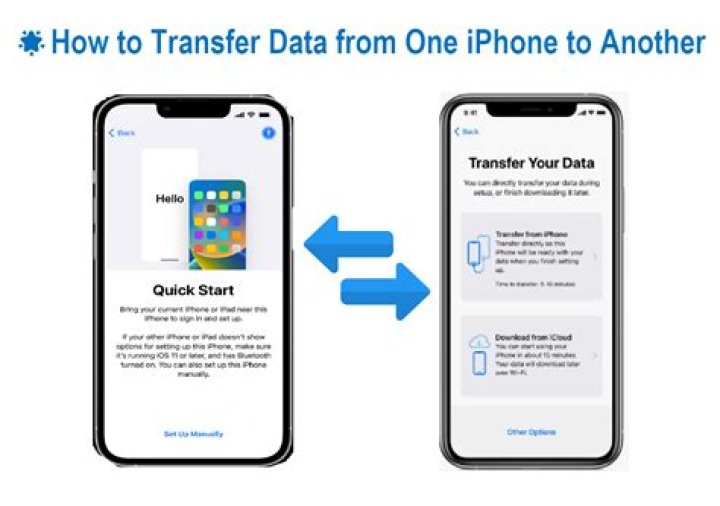
Isotopes are atoms with the same number of protons but that have a different number of neutrons. Since the atomic number is equal to the number of protons and the atomic mass is the sum of protons and neutrons, we can also say that isotopes are elements with the same atomic number but different mass numbers..
Herein, how do you know which elements are isotopes?
Look up at the atom on the periodic table of elements and find out what its atomic mass is. Subtract the number of protons from the atomic mass. This is the number of neutrons that the regular version of the atom has. If the number of neutrons in the given atom is different, than it is an isotope.
what element has 4 isotopes? strontium
Also Know, is every element an isotope?
Yes, all elements have isotopes. An isotope is any variant of an element that has a given number of neutrons. There are plenty of elements that have only one naturally occurring isotope or only one isotope that occurs in more than trace amounts on Earth.
What causes an isotope?
The isotopes of an element are all the atoms that have in their nucleus the number of protons (atomic number) corresponding to the chemical behavior of that element. But since they have different numbers of neutrons, these isotopes of the same element may have different radioactivity.
Related Question Answers
Which element has highest isotopes?
cesium
How do you find electrons in an element?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).What is an isotope easy definition?
isotope. An isotope of a chemical element is an atom that has a different number of neutrons (that is, a greater or lesser atomic mass) than the standard for that element. The atomic number is the number of protons in an atom's nucleus.How do you determine if an element is an ion?
The number of electrons is equal to the atomic number minus the charge of the atom. Refer to an element with either a positive or negative charge as an ion. The charge of the element should always be represented beside the symbol if it is an ion.How many elements are there?
118 elements
How many elements have no isotopes?
Which elements have no isotopes? All elements have two or more isotopes. You may be asking about elements with no stable isotopes, which include technetium (Z=43), promethium (Z=61), and everything from bismuth (Z=83) onward.What elements have no isotopes?
Elements that have no isotopes - Beryllium-9.
- Fluorine-19.
- Sodium-23.
- Aluminum-27.
- Phosphorus-31.
- Scandium-45.
- Manganese-55.
- Cobalt-59.
Can any element be an isotope?
Isotopes. Most atoms have several naturally occurring isotopes (click here for a list of elements that have no isotopes). An isotope is an atom which contains a different number of neutrons in its nucleus than some other atom of the same element. Not all isotopes are equally abundant in nature.Why is carbon 14 not an isotope?
Atoms of both isotopes of carbon contain 6 protons. Atoms of carbon-12 have 6 neutrons, while atoms of carbon-14 contain 8 neutrons. Although neutrons do not carry an electrical charge, they have a mass comparable to that of protons, so different isotopes have different atomic weight.What are 3 examples of isotopes?
Some examples of stable isotopes are isotopes of carbon, potassium, calcium and vanadium. Radioactive isotopes have an unstable combination of protons and neutrons, so they have unstable nuclei. Because these isotopes are unstable, they undergo decay, and in the process can emit alpha, beta and gamma rays.What is the mass number of an element?
The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom. Consider the table below, which shows data from the first six elements of the periodic table. Consider the element helium. Its atomic number is 2, so it has two protons in its nucleus.How do you find the mass number of an element?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.Why do all isotopes of an element have the same?
Atoms of the same element that differ in their numbers of neutrons are called isotopes. Different isotopes of an element generally have the same physical and chemical properties because they have the same numbers of protons and electrons. Most hydrogen atoms lack a neutron and are just called hydrogen.What is an isotope symbol?
Isotope notation, also known as nuclear notation, is important because it allows us to use a visual symbol to easily determine an isotope's mass number, atomic number, and to determine the number of neutrons and protons in the nucleus without having to use a lot of words. Additionally, N=A−Z.What is an isotope example?
Isotope Examples Carbon 12 and Carbon 14 are both isotopes of carbon, one with 6 neutrons and one with 8 neutrons (both with 6 protons). Carbon-12 is a stable isotope, while carbon-14 is a radioactive isotope (radioisotope). Uranium-235 and uranium-238 occur naturally in the Earth's crust.How many electrons does oxygen have?
2, 6
What does the 14 in carbon 14 represent?
The 14 in carbon-14 represents the mass number of the carbon-14 isotope. The mass number is the total count of both the protons and neutrons in the nucleus of the atom. Since all carbon atoms have six protons, only the number of neutrons will change from isotope to isotope.Which is the most stable element?
The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold.Is hydrogen an isotope?
(The word isotope refers to a nucleus with the same Z but different A). There are three isotopes of the element hydrogen: hydrogen, deuterium, and tritium. Hydrogen has no neutron, deuterium has one, and tritium has two neutrons. The isotopes of hydrogen have, respectively, mass numbers of one, two, and three.