The catalase test tests for the presence of catalase, an enzyme that breaks down the harmful substance hydrogen peroxide into water and oxygen. If an organism can produce catalase, it will produce bubbles of oxygen when hydrogen peroxide is added to it. Add one drop of hydrogen peroxide and look for bubbles..
People also ask, what is the purpose of the catalase test?
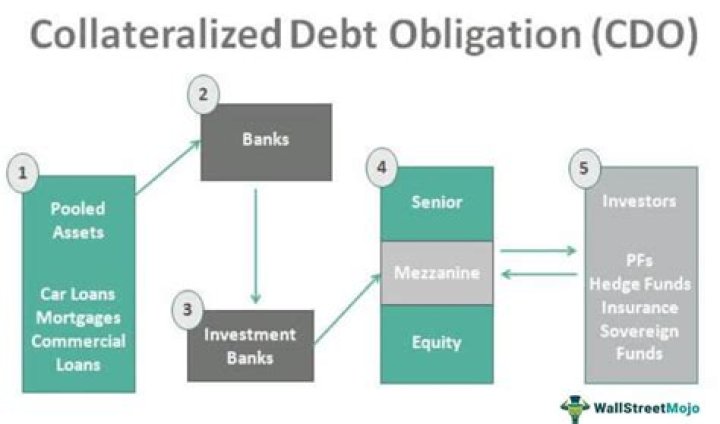
The catalase test is used to differentiate staphylococci (catalase-positive) from streptococci (catalase-negative). The enzyme, catalase, is produced by bacteria that respire using oxygen, and protects them from the toxic by-products of oxygen metabolism.
Likewise, what does it mean to be catalase positive? Catalase is an enzyme that converts hydrogen peroxide to water and oxygen gas. The test is easy to perform; bacteria are simply mixed with H 2O 2. If bubbles appear (due to the production of oxygen gas) the bacteria are catalase positive. If no bubbles appear, the bacteria are catalase negative.
Also Know, how does a catalase test work?
The catalase test tests for the presence of catalase, an enzyme that breaks down the harmful substance hydrogen peroxide into water and oxygen. If an organism can produce catalase, it will produce bubbles of oxygen when hydrogen peroxide is added to it. Bubbles are a positive result for the presence of catalase.
Is bacillus catalase positive or negative?
Catalase test is used to differentiate aerotolerant strains of Clostridium, which are catalase negative, from Bacillus species, which are positive.
Related Question Answers
What would happen if catalase stopped working?
If hydrogen peroxide is not broken down by catalase, additional reactions convert it into compounds called reactive oxygen species that can damage DNA, proteins, and cell membranes. A shortage of this enzyme can allow hydrogen peroxide to build up to toxic levels in certain cells.Where is catalase found?
liver
Why catalase test is important?
Semiquantitative catalase test is used for the identification of Mycobacterium tuberculosis. It is used to differentiate aerotolerant strains of Clostridium, which are catalase negative, from Bacillus species, which are positive. Catalase test can be used as an aid to the identification of Enterobacteriaceae.What types of bacteria are catalase positive?
List of catalase positive microorganisms - Staphylococci.
- Pseudomonas aeroginosa.
- Aspergillus fumigatus.
- Candida albicans.
- Enterobacteriaceae (Klebsiella, Serratia)
- Mycobacterium tuberculosis produces a heat-labile catalase workable only at body temperatures.
What does hydrogen peroxide do?
Hydrogen peroxide is a mild antiseptic used on the skin to prevent infection of minor cuts, scrapes, and burns. It may also be used as a mouth rinse to help remove mucus or to relieve minor mouth irritation (e.g., due to canker/cold sores, gingivitis).What reagent is used to perform a catalase test?
To observe the action of these enzymes, catalase reagent, a dilute solution of hydrogen peroxide, is added to a pure bacterial culture. Any immediate bubbling is indicative of a positive result since oxygen is a byproduct of hydrogen peroxide decomposition.Does E coli produce catalase?
Catalase is a ubiquitous antioxidant enzyme that degrades hydrogen peroxide into water and oxygen1. Escherichia coli has 2 catalase enzymes, hydroperoxidase I (HPI) and HPII, which catalyze the dismutation of hydrogen peroxide to water and oxygen1.Why do obligate anaerobes not require catalase?
Obligate anaerobes, which live only in the absence of oxygen, do not possess the defenses that make aerobic life possible and therefore cannot survive in air. One study showed that facultative and aerobic organisms lacking superoxide dismutase possess high levels of catalase or peroxidase.How can you tell that catalase has been added to hydrogen peroxide?
How can you tell that catalase has been added to hydrogen peroxide? Oxygen bubbles will form, and that's when one can tell that catalase has been added.How fast is catalase?
Thus, the catalases constitute some of the most efficient enzyme systems known, with large specificity constants kcat/Km of around 108 to 109 M-1 s-1 [Web reference 1]. This means catalases are capable of disproportionating many tens of millions of hydrogen peroxide (H2O2) molecules per second.What would happen if you increase the enzyme concentration?
By increasing the enzyme concentration, the maximum reaction rate greatly increases. Conclusions: The rate of a chemical reaction increases as the substrate concentration increases. Enzymes can greatly speed up the rate of a reaction. However, enzymes become saturated when the substrate concentration is high.Is E coli positive for catalase test?
Escherichia coli and Streptococcus pneumoniae have been used as model catalase-positive and catalase-negative bacteria, respectively. The reusability of the biosensor was improved by placing a nylon membrane on the bioelectrode surface to prevent fouling caused by the bacterial medium.Why is Streptococcus catalase negative?
Staphylococci are catalase positive whereas Streptococci are Catalase negative. Catalase is an enzyme used by bacteria to induce the reaction of reduction of hydrogen peroxide into water and oxygen.What contains catalase?
There are tons of vegetable sources of catalase: Leeks, onions, broccoli, parsnips, zucchini, spinach, kale, radishes, carrots, red peppers, turnips, cucumbers, celery, avocado, potato, and red cabbage all have rich stores of catalase.Which bacteria are oxidase positive?
Oxidase Positive Organisms: Pseudomonas, Neisseria, Alcaligens, Aeromonas, Campylobacter, Vibrio, Brucella, Pasteurella, Moraxella, Helicobacter pylori, Legionella pneumophila, etc.Does catalase reverse gray hair?
Researchers recently discovered that going gray is a buildup of hydrogen peroxide in your hair particles, which bleaches your hair from the inside. Usually an enzyme called catalase breaks down hydrogen peroxide into water and oxygen, but as you get older, catalase production starts to slow down.Why is catalase important in the human body?
Catalase is a common enzyme found in nearly all living organisms exposed to oxygen (such as bacteria, plants, and animals). It catalyzes the decomposition of hydrogen peroxide to water and oxygen. It is a very important enzyme in protecting the cell from oxidative damage by reactive oxygen species (ROS).What affects catalase?
The rate at which an enzyme works is influenced by several factors including the concentration of substrate (hydrogen peroxide in the case of catalase), temperature, pH, salt concentration and the presence of inhibitors or activators. Every enzyme has an optimal range for each of these factors.Are enzymes proteins?
Enzymes are biological molecules (proteins) that act as catalysts and help complex reactions occur everywhere in life. Let's say you ate a piece of meat. Proteases would go to work and help break down the peptide bonds between the amino acids.