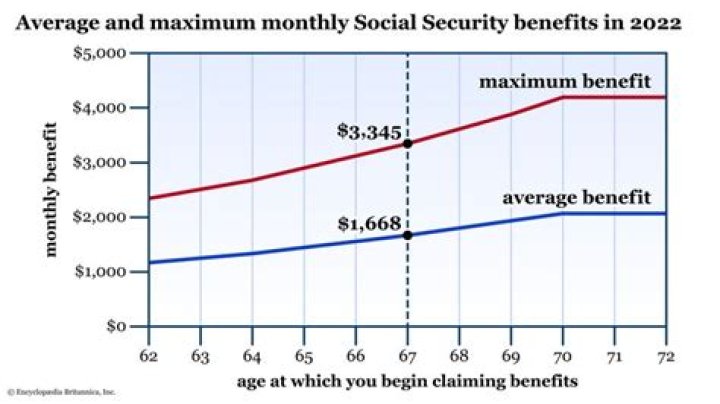
.
Also know, why does the third shell have 8 electrons?
The third shell in its lowest state has room for 8 electrons but including the higher energy 3d electrons it has a capacity of 18 electrons.
Likewise, how many electrons are allowed in the 4th Shell? The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons.
Also Know, why does the third shell of an atom have 8 electrons when it has the capacity of 18 electrons?
The electron capacity of the third shell is 8, when there are no shells above it. And that is the case for all elements in the third period. It is only when there are outer shells surrounding it that the third (or higher) shell has a higher capacity. (The number of electrons in the nth energy level = 2n².
How many valence electrons are in the third shell?
5 valence electrons
Related Question AnswersWhy can there only be 8 valence electrons?
This is because of the quantum nature of the atoms, where electrons are organized in shells: the first (named the K shell) has 2 electrons, the second (L-shell) has 8, the third (M shell) has 18. Atoms combine into molecules by trying in most cases to have valence electrons entirely filling a shell.Why are there only 8 electrons in the outer shell?
The eight- electrons stability of an atom is stemmed from the stability of the noble gases or the elder name inert gases, which had long been known as unreactive or noble. However, this rule is justified for elements of second row in the periodic table, which their outermost- shell capacity is 8 electrons.Can there be more than 8 electrons in a shell?
Some molecules can accommodate more than 8 valence electrons because they have expanded valence shells. These molecules are from periods 3 or higher because starting from n=3, atoms have d-orbitals that can accommodate for more that 9 valence electrons.How many electrons can 3p hold?
six electronsWhy are there 8 electrons in the outer shell?
Row 2 elements on the left will lose electrons to get a full outer shell, elements on the right will gain electrons to get 8 electrons in their outer shell. The basis of the octet rule is that atoms 'want' a full outer shell of electrons, i.e. they are most stable with a full outer shell.How is Valency calculated?
The valency of an atom is equal to the number of electrons in the outer shell if that number is four or less. Otherwise, the valency is equal to eight minus the number of electrons in the outer shell. Once you know the number of electrons, you can easily calculate the valency.Why does calcium only have 8 electrons in the third shell?
Why are there 8 electrons in the third shell of calcium instead of 10? This principle states that, “Electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. In this way, the electrons of an atom or ion form the most stable electron configuration possible.”Why do shells start with K?
He noticed that atoms appeared to emit two types of X-rays. As it turns out, the K type X-ray is the highest energy X-ray an atom can emit. It is produced when an electron in the innermost shell is knocked free and then recaptured. This innermost shell is now called the K-shell, after the label used for the X-ray.What is the capacity of M shell?
The L shell only holds eight electrons. The M shell only holds eight electrons. The M shell can actually hold up to 18 electrons as you move to higher atomic numbers. The maximum number of electrons you will find in any shell is 32.How do electrons fill in orbitals?
RULES FOR FILLING ORBITALS. Rule 1 - Lowest energy orbitals fill first. Thus, the filling pattern is 1s, 2s, 2p, 3s, 3p, 4s, 3d, etc. Since the orbitals within a subshell are degenerate (of equal energy), the entire subshell of a particular orbital type is filled before moving to the next subshell of higher energy.What is Subshell?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.How do you distribute electrons in shells?
According to Bohr Bury Scheme, the electronic distribution in the first four shells is: K(2), L(8), M(18), N(8). Outermost shell can not have more than 8 electrons and K-shell can have a maximum of two electrons. If atomic number of an element is known, arrangement of electrons in its atom can be written.How many Subshells are in the N 3 shell?
three subshellsHow many Subshells are in the 4th Shell?
Answer: There are 4 subshells: 4s, 4p, 4d, and 4f. When the question asks you about how many subshells there are in a certain shell, what it basically is asking is how many different values of l there are. So if n=4, l can only be 0,1,2,3, which corresponds to s,p,d,f.How many electrons can the 7th Shell hold?
Notice that you can easily find the number of electrons that an atom can accomodate on a particular shell, or energy level, by using the fact that each orbital can hold a maximum of two electrons.How many electrons are in 4s?
two electronsHow many electrons can 5f hold?
Each orbital can hold two electrons, so the capacity of an nf subshell is 14 electrons.How many electrons are in the 4th shell of ytterbium?
Ytterbium atoms have 70 electrons and the shell structure is 2.8.How do you find the quantum number of electrons?
How to Determine the Number of Electrons With Quantum Numbers- Count the Full Orbitals.
- Add the Electrons for Each Full Orbital.
- Identify the Subshell Indicated by the Angular Quantum Number.
- Add the Electrons from the Full Subshells.
- Add the Electrons from Full Subshells to Those From Full Orbitals.
- Find the Legitimate Vales for the Magnetic Quantum Number.